Header Picture: Carcharodontosaurus saharicus tooth - © Jurassic-Dreams.com
We get used to the wonderful colors of some fossils, as if they were magic, featuring a range of bright and beautiful tonalities. But it is not magic... And science has the answer.
What Geological Phenomenon Creates Those Enamel Hues?
There are a series of processes that occur since the animal dies until its remains come to our hands. And this is relatively easy to explain putting as example the dinosaur teeth.
Biological remains may undergo alterations of their original chemical and mineralogical composition when incorporated into the fossil record. These processes continue after the burial and even after the collection of the fossil.
To analyze the origin of the color in the dinosaur teeth, first we must understand the structural configuration and the original composition.
Two Main Layers: Enamel and Dentin
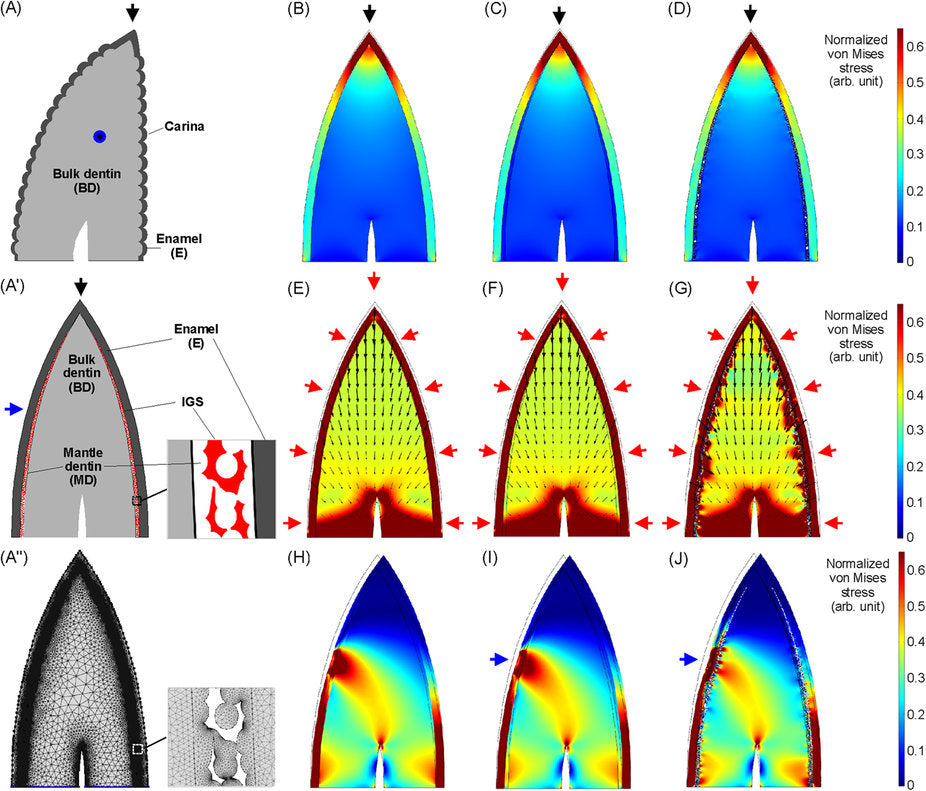
Structure of a Saurischian Tooth, Source: Nature Scientific Report
In the following Spinosaurus tooth, we can see the different layers exposed, which give us an educational insider perspective on the fossil. The tooth's layers are visible because of the erosion, which are a product of ancient transport processes during the sedimentation.

Visible layers in a Spinosaurus tooth - © Jurassic-Dreams.com
Obviously nobody was there, 95 million years ago, to analyze the composition of a dinosaur tooth. However, in most studies it is premised that the original composition would have been similar to the teeth of the present vertebrates, mainly composed of the mineral hydroxyapatite (hydrated calcium phosphate).
It is easy to understand that usually the teeth did not have any type of pigmentation during the life of the animal. However, during their burial and before the sediments underwent a complete lithification, many physical-chemical processes occurred, that altered the original composition of the remains.
Mineralization
It is mainly due to the fluids that circulated between the sediments before their lithification. These aqueous fluids sometimes had high concentrations of certain elements which interacted with each other and with the medium, causing great mineralogical changes in the processes of sedimentary petrogenesis.
The mineralization of the preserved elements can be by addition of new mineral components (cementation) or by substitution of the minerals existing in them (neomorphism or replacement).
Minerals With Greatest Influence
The main minerals that have intervened in the mineralization of the preserved elements, and currently found in fossils, are:
- Calcite (calcium carbonate)
- Dolomite (magnesium carbonate)
- Pyrite and marcasite (iron sulfide)

- Limonite (hydrated iron oxide)
- Hematite (iron oxide)
- Opal (hydrated siliceous oxide)

Opalized ammolite, by Kumiko, CC By-SA 2.0
- Gypsum (hydrated calcium sulphate)
- Glauconite (hydroxy-silicate of Fe, Al, Mg)
- Vivianite (hydrated iron phosphate)
- Apatite and hydroxyapatite (calcium phosphates)
- Quartz (silicon oxide)

Silicified wood from the African Mesozoic
In some exceptional sites, fossils consisting of baritine, uraninite, and other radioactive minerals. Siderite, rhodochrosite, chalcopyrite, diopside or tremolite are also known. And yet rarer are the fossils constituted by azurite, malachite, anglesite, fluorite, galena, blenda, or native metals like silver and copper.
Durability
The durability of the preserved elements depends on the stability of their mineral components. In general, the mineralization of the preserved elements increases their physical resistance. However, other properties such as chemical or mineralogical composition, micro-structure, shape, size and thickness of some elements are also modified.
Change of Composition
The process of alteration of teeth originally composed by hydroxyapatite is a replacement. This taphonomic process consists of the substitution of an original mineral component for another of different chemical composition. The modifications experienced by a rest subjected to this process may be very small and affect only the composition of the trace elements. The micro-structure of the affected remains usually persists when the replacement takes place molecule to molecule.
Stability
The hydroxyapatite of the teeth is thermodynamically unstable and usually undergoes modifications at the same time that the organic substances are degraded (Fernández López, 2000). Mineral species of the apatite group that have low fluorine concentrations tend to be transformed into fluorine rich species during fossilization.
Due to this fluorination the rests become more resistant, giving rise to fluorapatite, a more stable phosphate with larger crystalline domains and therefore less soluble (Moreno-Azanza, M., et al., 2013).
However, these changes to more stable forms of apatite do not alter the color, simply chemically stabilizing it. Here the trace elements present in the structure of the hydroxyapatites and the elements that are in high concentration in the sedimentary environment come into play.
Case Study: The Red-Orange-Yellow From the Sahara
The Ifezouane and Aoufous Formations of the Cenomanian stage (Upper Cretaceous) from the South of Morocco and Algeria, possess a large amount of sedimentary iron in the form of oxides and hydroxides (limonite), which is characterized by strong red, orange and yellow tonalities (remember the typical orange color of a rusty iron object).
The porous structure of the teeth dentin favored the process of alteration, where the metal ions were incorporated into the molecular structure of fluorapatite, as well as crystallizing as oxides in the porosity and structural microcracks of the tooth. The high availability of iron in these sandstone formations boosted the genesis of calcium fluorophosphate with high iron concentrations.
For this reason, most of the teeth that appear preserved in this geological context have a nice coloration of their enamel and dentin. The variations between the tonalities depend on the concentration of iron present and its oxidation state. Black and very strong reddish teeth usually possess a large amount of iron oxide in its internal structure.
Visual Examples From White to Grey
Illustrative examples of dinosaur teeth from the Ifezouane and Aoufous Formations that present different concentrations of iron in their structure, featuring a diversity of enamel tonalities as a consequence.
White/Light Yellow (low concentration of iron oxide)
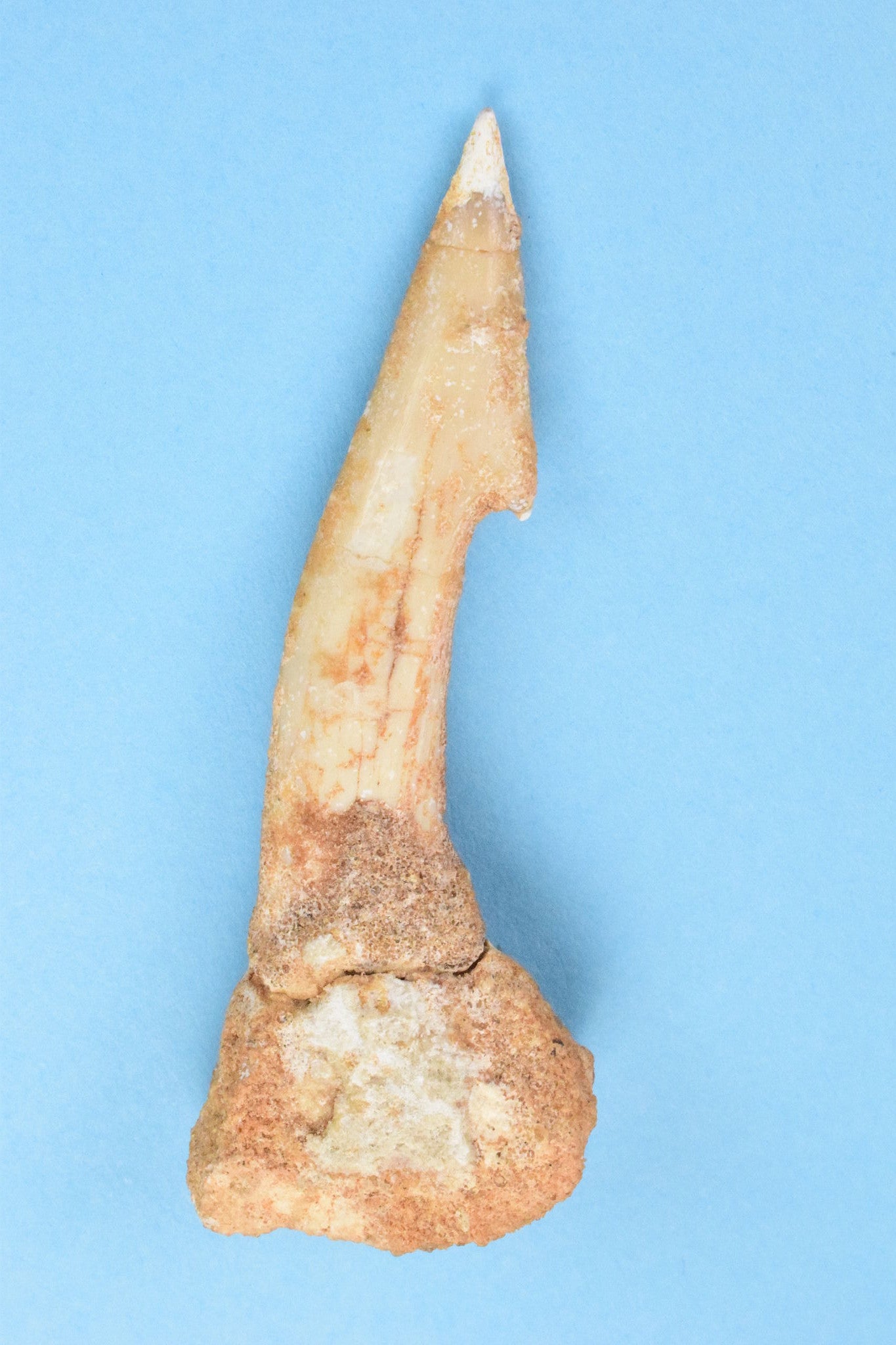
Onchopristis numidus tooth © Jurassic-Dreams.com
Yellow (medium-low concentration of iron oxide)

Dromaeosaur Raptor tooth © Jurassic-Dreams.com
Brown (medium concentration of iron oxide)

Onchopristis numidus tooth © Jurassic-Dreams.com
Orange/Light Red (medium-high concentration of iron oxide)

Carcharodontosaurus saharicus tooth © Jurassic-Dreams.com
Red/Dark Red (high concentration of iron oxide)

Spinosaurus aegyptiacus tooth © Jurassic-Dreams.com
Grey (mineralization of iron oxide through microcracks)

Dromaeosaur Raptor tooth © Jurassic-Dreams.com
Beyond the Color Curiosity. A Real Scientific Value
Apart from the decorative value of teeth with showy mineralizations, what scientific information of great value can be extracted from the enamel and dentin of the dinosaur teeth?
The enamel of a dinosaur tooth can give us information about the climate of the moment in which it lived. A study of the Carnegie Institution showed that "the key to infer past temperatures [...] is in measuring small differences in the chemical composition of oxygen in dinosaur tooth enamel", because "dinosaurs drink and ingest water from the streams, [...] then incorporate oxygen from this water into their tooth enamel". And the oxygen isotope ratio remains constant, so we can deduce the temperature today.
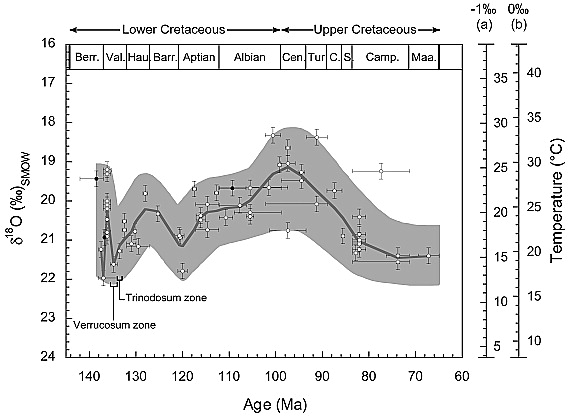
Temperature estimations based on fish tooth enamels, Source: Paleocenography Journal
Last but not least, cladistic analysis is possible by looking at the dental encimei. This basically means that the enamel also facilitates the taxonomical classification.
Not so bad for "the cover" of a fossilized tooth. If the enamel alone can provide us all this information, imagine when we have complete articulated rests, skulls or full jaws.
References:
- Fernández López, 2000: "Themes of Taphonomy". Paleontology Department, Geological Sciences Faculty, Complutense University of Madrid.
- Moreno-Azanza, M., et al., (2013): "Historia de un diente de Dinosaurio: Evolución mineralógica y tafonómica". Aragosaurus-IUCA Group, Earth Sciences Faculty, Zaragoza University